- Blog
- Kindergarten 2 game
- Pokemon omega ruby citra rom download
- Mixed in key download free pc
- Happy wheels full version free download
- Cricut betty boop svg free
- Least ads best solitaire classic for android
- Website manga hentai
- Retroarch ps2 border
- Commercial sewage ejector pump systems
- Call of duty 3 free download torrents
- Free hebrew script font
- City car driving games free download
- Reiboot pro for ipad
- Save wizard license key free 2021
- Download 8 ball pool miniclip fullscreenn
- Oatmeal raisin cookies recipe
- Bill of particulars new jersey
- Periodic table molar mass cl2
- Rfactor 2 track builder
- Scrivener 3 themes windows
- Monthly expense google sheet template
- Blue among us app logo
- Gta 6 game video
- Eagles eye lccc
- Cavalier king charles spaniel rescue sc
- Minecraft resource packs xray 1-14-4
- Europe time zone map
- Resume templates
- Minecraft steve smash release date
- Sims 3 complete download free
- Dust bowl fireside chats
- Unblocked games happy wheels full version free
- Long bodied cellar spider facts-
- Dirty text art copy and paste
- Xbox 360 emulator v3-2 download
- Us address mail forwarding service
- Superpowered tamara walkthrough
- Agent daily schedule pdf
- Ds 160 online form india
- Recording with vlc media player
- Family scary movies on netflix
- Is impaired physical mobility a nursing diagnosis
- Install google chrome on linux
- St petersburg fl party planners
- Flight 624 jfk arrival time
- Usps passport scheduler grand junction
- Commercial real estate flyer design
- Periodic table with exact molar mass
- Anchor chart story elements plot
:max_bytes(150000):strip_icc()/GettyImages-175532236-c614b233b7e84d5487cad8b280f365a4.jpg)
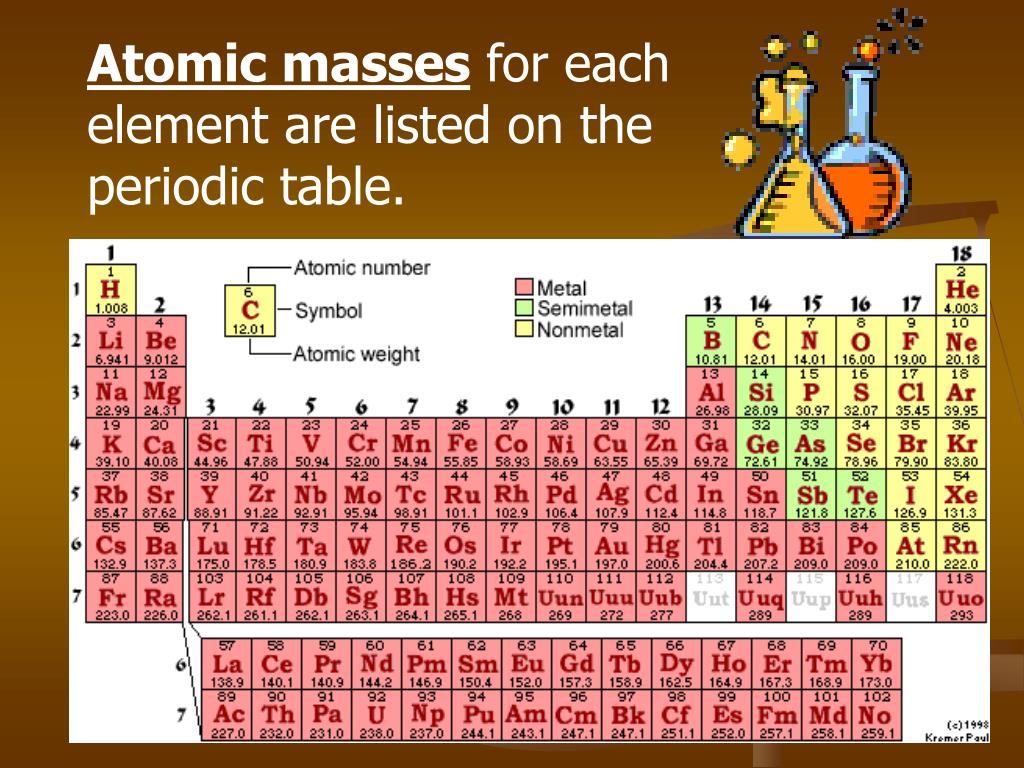
To use this resource Click Here.Ī useful resource, written by Jef Rozenski, calculates all possible combinations of H, C, N & O that give a specific nominal mass. Of course, compounds of chlorine and bromine have very large isotope abundances.Ī more elegant and complete isotope pattern calculator has been created by Junhau Yan. The numbers displayed in the M+1 and M+2 boxes are relative to M being set at 100%. Simply enter an appropriate subscript number to the right of each symbol, leaving those elements not present blank, and press the " Calculate" button. The calculator on the left may be used to calculate the isotope contributions to ion abundances 1 and 2 amu greater than the molecular ion (M). For compounds of chlorine and bromine, increments of 1.997 and 1.998 respectively must be added for each halogen to arrive at the higher mass isotope values. The molecular mass of a substance is defined as the sum of atomic masses of all the atoms in a molecule of a substanceThe molecular mass of Cl 2 2 × a t o m i c m a s s o f c h l o r i n e 2 × 35. Only the mass of the most abundant isotope, relative to C (12.0000), is used for these calculations. The mass calculator on the right may be used to calculate the exact mass of a molecule based on its elemental composition. This table is adapted from Introduction to Mass Spectrometry, by J.T. Visualize trends, 3D orbitals, isotopes, and mix compounds. You can see that The molar mass of Zinc is 65.38 g/mol. Interactive periodic table showing names, electrons, and oxidation states. So let’s look at the molar mass of Zinc and Chlorine from the above periodic table. If you know how to calculate molar mass, learn about other ways to express the amount of.

Find the molar mass of glucose by multiplying the atomic masses of the atoms and their number, then find the sum: 6 × 12.01 g/mol + 12 × 1.0079 g/mol + 6 × 16 g/mol 180.1548 g/mol. Now in ZnCl2, there is 1 Zinc atom and 2 Chlorine atoms. Count the number of atoms of each element in the compound. The factor is multiplied by the number of atoms (n) of the designated element to calculate the intensity contribution from higher mass isotopes. You can see the molar mass value of all the atoms from this periodic table. * X represents the relative intensity of the lowest mass ionin an isotopic ion cluster. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). And don’t forget to put the unit g/mol to your final calculated molar mass.Masses Exact Masses & Isotope Abundance Ratios Element The molar mass of Cl2 (Chlorine) is: 70.9 grams/mol.First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation. The molar mass is used to convert grams of a substance to moles and is used often in chemistry.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. I hope you have understood the short and simple calculation for finding the molar mass of Cl2. Prepare a concept map and use the proper conversion factor. Hence the Molar mass of Cl2 is 70.9 g/mol. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in (2Cl2): Molar Mass (g/mol) Cl (Chlorine) 4 × 35.453 141.812. So, Molar mass of Cl2 = Molar mass of 2 Chlorine (Cl) atoms. You can see that in Cl2, there are 2 Chlorine atoms. Now, to calculate the molar mass of Cl2, you just have to add the molar mass of all the individual atoms that are present in Cl2. The molar mass of Chlorine is 35.45 g/mol. So let’s look at the molar mass of Chlorine from the above periodic table. You can see the molar mass value of all the atoms from this periodic table.
- Blog
- Kindergarten 2 game
- Pokemon omega ruby citra rom download
- Mixed in key download free pc
- Happy wheels full version free download
- Cricut betty boop svg free
- Least ads best solitaire classic for android
- Website manga hentai
- Retroarch ps2 border
- Commercial sewage ejector pump systems
- Call of duty 3 free download torrents
- Free hebrew script font
- City car driving games free download
- Reiboot pro for ipad
- Save wizard license key free 2021
- Download 8 ball pool miniclip fullscreenn
- Oatmeal raisin cookies recipe
- Bill of particulars new jersey
- Periodic table molar mass cl2
- Rfactor 2 track builder
- Scrivener 3 themes windows
- Monthly expense google sheet template
- Blue among us app logo
- Gta 6 game video
- Eagles eye lccc
- Cavalier king charles spaniel rescue sc
- Minecraft resource packs xray 1-14-4
- Europe time zone map
- Resume templates
- Minecraft steve smash release date
- Sims 3 complete download free
- Dust bowl fireside chats
- Unblocked games happy wheels full version free
- Long bodied cellar spider facts-
- Dirty text art copy and paste
- Xbox 360 emulator v3-2 download
- Us address mail forwarding service
- Superpowered tamara walkthrough
- Agent daily schedule pdf
- Ds 160 online form india
- Recording with vlc media player
- Family scary movies on netflix
- Is impaired physical mobility a nursing diagnosis
- Install google chrome on linux
- St petersburg fl party planners
- Flight 624 jfk arrival time
- Usps passport scheduler grand junction
- Commercial real estate flyer design
- Periodic table with exact molar mass
- Anchor chart story elements plot
